Highly Validated Approaches to Highly Unmet Needs.
Developing Treatments for Severe Liver Disease.
about
Inipharm is a clinical-stage biopharmaceutical company focused on discovering and developing therapies for severe liver diseases. Inipharm's lead program is focused on the highly validated, genetically-defined target, HSD17B13.
Inipharm, through its Australian subsidiary Inipharm Australia, Pty Ltd, is conducting a Phase 1 clinical trial, including MASH patients, of INI-822, a first in class small molecule inhibitor of HSD17B13.
There is extensive, consistent evidence that genetic variants of HSD17B13 are associated with significantly lower rates and severity of multiple liver diseases. Building on these novel insights into the biologic activity of HSD17B13, Inipharm is advancing a pipeline of small-molecule therapies that target the activity of this protein.
clinical development
First clinical-stage small molecule inhibitor of HSD17B13
Inipharm, through its Australian subsidiary Inipharm Australia, Pty Ltd, is conducting clinical development of INI-822, the first small molecule HSD inhibitor. Phase 1 work in healthy volunteers has been completed and testing in patients with MASH is underway. For more information:
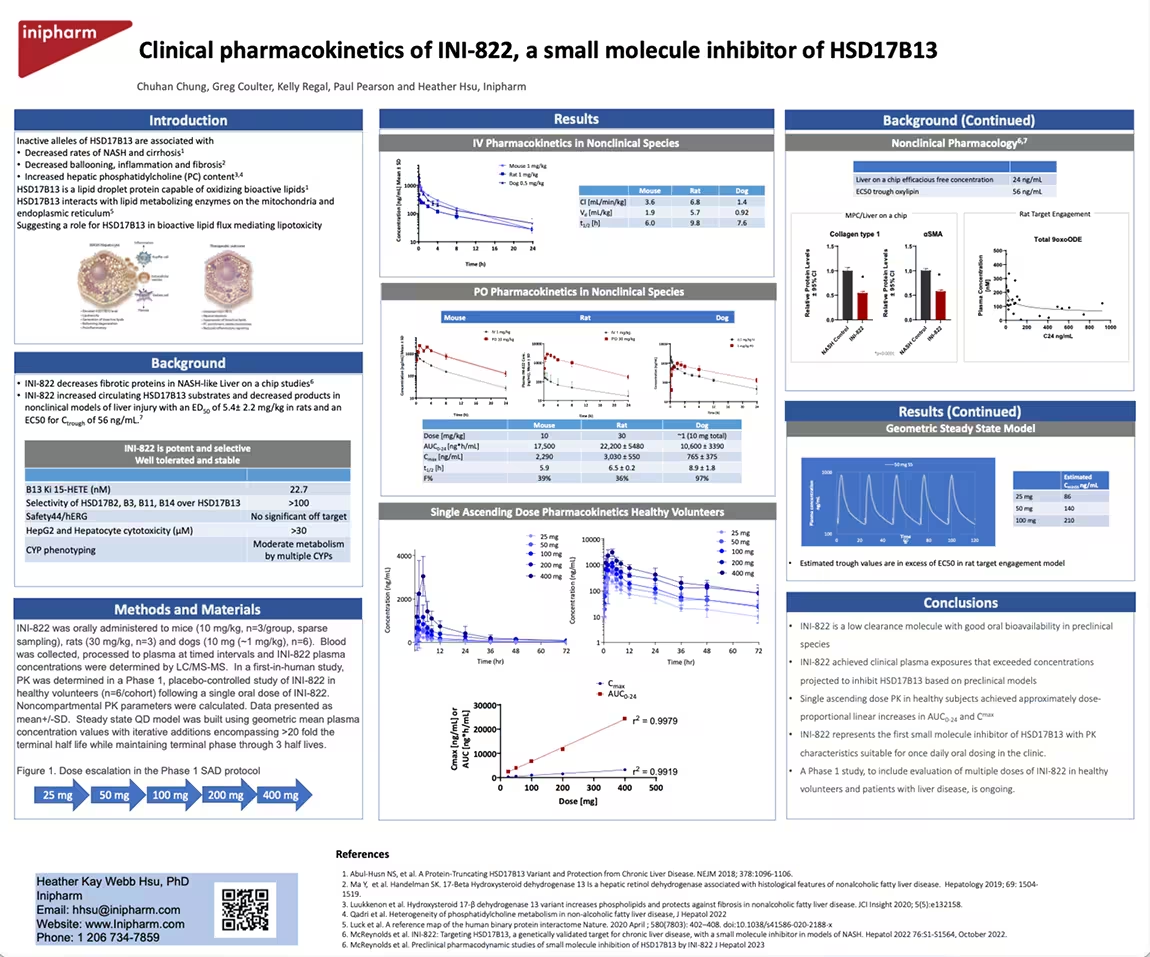
Clinical pharmacokinetics of INI-822, a small molecule inhibitor of HSD17B13
our science
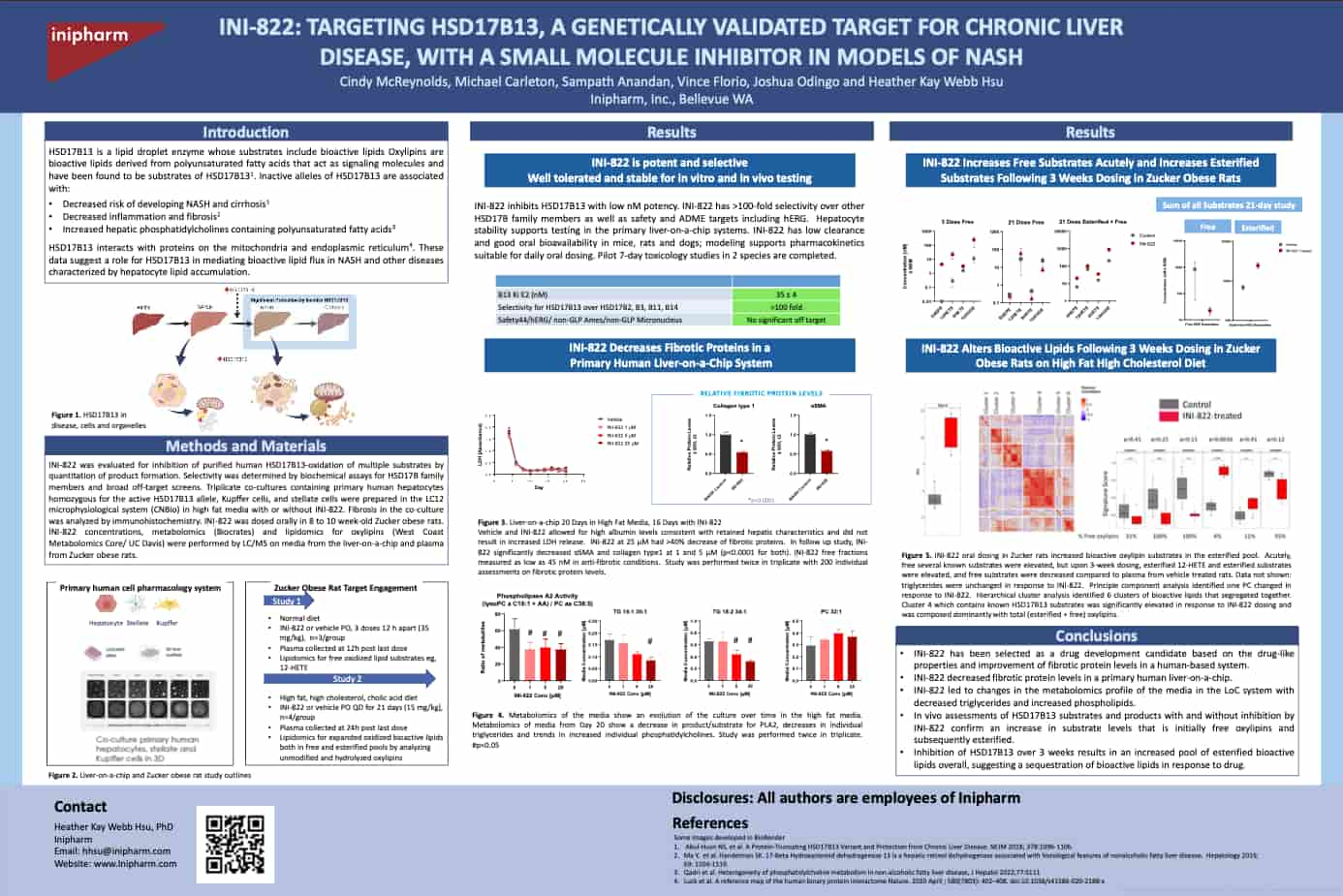
Inipharm is developing small-molecule inhibitors of HSD17B13 to treat fibrotic liver diseases. Inipharm's inhibitors have been shown to drive alterations in lipid metabolism and improvements in liver enzymes that are consistent with changes seen with the protective variant in humans. In human liver cell-based systems, INI-822 demonstrated anti-fibrotic effects.
Protective variants in HSD17B13 are associated with reduced risk incidence and/or the severity of numerous liver diseases.
- HSD17B13 has been identified as a novel target for NASH and driver of liver disease from a genome wide association study.
- Loss of function variants in HSD17B13 are associated with reduced risk incidence and/or the severity of numerous liver diseases.
- Importantly, HSD17B13 has been shown to be involved in pathways associated with lipotoxicity, inflammation, and fibrosis.

INI-822, development candidate
- Reduced fibrotic markers in human liver cell model
- Improved liver enzymes in multiple in vivo models
- Replicated lipidomic changes seen with protective HSD17B13 variant
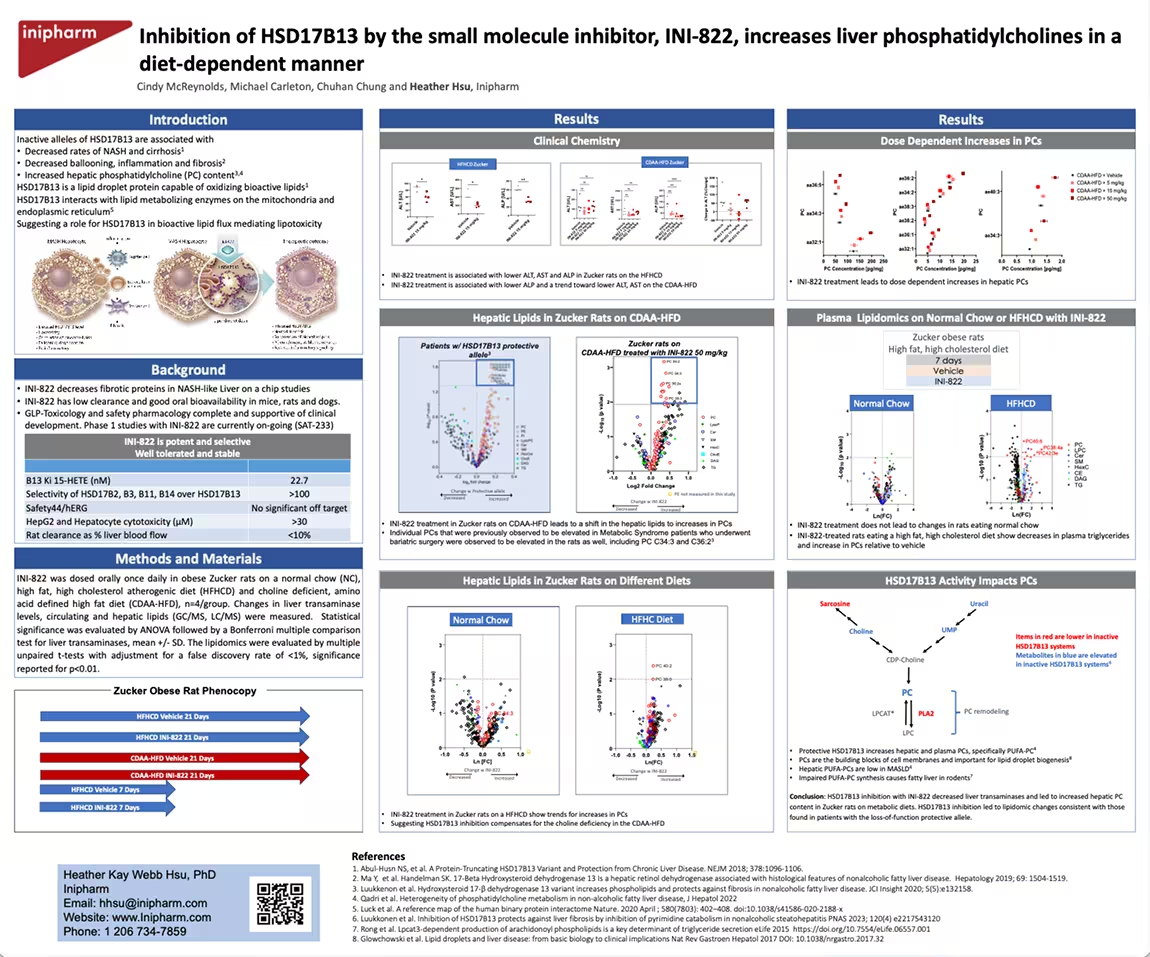
Inhibition of HSD17B13 by the small molecule inhibitor, INI-822, increases liver phosphatidylcholines in a diet-dependent manner
leadership
board of directors
scientific advisory board
Inipharm Australia Pty Ltd board of directors
Mario Pennisi, AM
Director
news and publications
- June, 2026 Inipharm Initiates Dosing in 12-Week Proof-of-Concept Clinical Study in MASH With Its Oral HSD17B13 Inhibitor INI-822
- May, 2024 Inipharm to Present Pharmacokinetic Data From a Phase 1 Clinical Trial of INI-822, a Small Molecule Inhibitor of HSD17B13
- November 30, 2023 Inipharm Initiates Dosing in Phase 1 Study of Its Small Molecule Inhibitor of HSD17B13
- June 7, 2023 Inipharm's Development Candidate INI-822 Shows Improvements in Markers of Liver Homeostasis in Preclinical Studies
- October 25, 2022 Inipharm to Present Data on Anti-Fibrotic Effects of Its Development Candidate Targeting HSD17B13 at AASLD’s The Liver Meeting
- June 8, 2022 Inipharm Appoints Chuhan Chung, M.D, as Chief Medical Officer to Advance HSD17B13 Program into the Clinic
- October 05, 2021 Inipharm to Present Data Showing Potential of Small Molecule Inhibitors of HSD17B13 to Combat Liver Fibrosis at AASLD's The Liver Meeting
- November 11, 2020 Inipharm Raises $35 Million in Series A to Focus on Highly Validated Genetic Target HSD17B13

